Статьи по темам
Фотогалерея
Intrinsic sustained activity of bicarbonate aqueous solutions
2009 г.
*Voeikov V.L., Do Ming Ha, Mukhitova O.G., Vilenskaya N.D., Malishenko S.I., Bogachuk A.S.
*Lomonosov Moscow State University, Faculty of Biology, Moscow, Russia
The major source of energy for the performing all vital functions is respiration. The current concept of aerobic respiration restricts oxygen consumption to mitochondrial respiration, due to which ATP is produced considered to secure all energy requirements of an organism. Oxygen is used here at the end of the electron-transport chain as a final acceptor of “exhausted” electrons. Half a century ago Albert Szent-Gyorgyi suggested the existence of an alternative energy-production system in which “hot” electrons are directly transferred to O2 [1]. Sequential one-electron oxygen reduction to water [O2 + (4e─ + 4H+) -> 2H2O] is accompanied with the release of big quanta of energy (high density energy) able to support living matter in a highly excited state. Such energy production is a kind of genuine combustion or “burning”. There is evidence that a significant part of oxygen consumed by animals and plants is used in such a way.
Generally combustion is not regarded as relevant for bioenergetics. One of the reasons for this is that it is commonly considered that combustion can not efficiently proceed in water constituting the major material component of living matter. However, it has been proved very long ago that water is necessary for any combustion though no mechanism of it participation has been suggested [2].
Currently evidence accumulates that two different aqueous phases are present in liquid water – domains with quasi-liquid crystal structure and “gas” – like water. Water molecules reside in organized domains in a coherent excited state, and this phase has electron donating (reducing) properties [3]. The preferred acceptor of electrons is oxygen that is always present in water. Under certain conditions a chain reaction of oxygen reduction may be initiated. Essentially this process represents slow water burning. Its peculiarity is in apparent identity of reagents and final reaction products:
O2 + 2H2O -> 2H2O + O2 + Energy
Nevertheless free energy is released in the course water burning as in the course of any other fuel combustion. Ultimately energy comes from the destruction of organized water phase due to “burning” of water belonging to it (left side of the equation). Right side of the equation indicates appearance of water in the ground state water phase. In principle, ground state water molecules may be recruited back into organized water phase providing for the stable functionality of this system.
It should be noted, however, that “burning” of organized water outlined in the equation shows some ideal situation. It probably can not realize in “pure” water. Certain catalysts are needed for the process of water “burning” to go efficiently. One of the “impurities” that may catalyze the processes related to water “burning” may be the family of carbonates:
CO2 <-> H2CO3 <-> HCO3─
They are commonly present in water because of wide occurrence of carbonates in nature and due to high solubility of CO2 in water. In fact it was proved long ago that normal respiration can not go without proper level of carbonates in blood, tissues and cells. According to Y. Henderson decrease of carbonates levels below some critical level especially in brain may result in fatigue and death due to respiration cessation [4]. Carbonates and in particular bicarbonates are active regulators of oxygen metabolism on a cellular level. They are also universal modulators of enzyme activity in vitro in particular of those enzymes that participate in direct oxygen reduction [5]. Inasmuch as significant part of consumed oxygen is one-electronically reduced, spent for combustion and since water in principle may be used as a fuel, it can not be excluded that carbonates catalyze this process, so that water containing carbonates may in a certain sense “respire”. If this is the case such aqueous systems can generate and accumulate high density energy.
Indeed, one can observe that a wave of photon emission (PE) amplified by luminol arises upon addition of salts of Fe(II) to bicarbonate aqueous solutions being in contact with air. Luminol is a well known as a luminescent probe for reactive oxygen species (ROS), intermediate products of one electron oxygen reduction and Fe(II) is a catalyst of peroxides decomposition. So PE indicates that spontaneous oxygen reduction proceeds in bicarbonate solutions contacting with the air. In its course metastable hydrogen peroxides (H2On, where n=2-4) may accumulate in the system making it persistently non-equilibrium and capable to release radiant energy in response to Fe(II) addition [6]. Recently we found that if H2O2 is added to weak bicarbonate solutions (1-5 mM) to final concentrations as small as 0,001-0,01%, these solutions turn into persistent source of luminol-amplified PE in the absence of added metal and even if these reaction systems were kept in the darkness in tightly closed vessels!. Some interesting peculiarities of such systems behavior suggest that they display properties of “respiring” systems.
Just after H2O2 injection to Na/KHCO3 solutions luminol-amplified PE could increase or decrease for 1-2 days. Then PE intensity stabilized around some mean level and did not disappear for months. Some substances significantly affect PE intensity even in ultra-low doses. Figure 1 illustrates the action of hydrated fullerenes C60 (HyFn) (Hydrated fullerenes (HyFn) is an aqueous solution of fullerene molecules С60, prepared in such a way that each fullerene molecule is enclosed in a multilayer aqueous shell. For such preparations pro- and antioxidant activity was demonstrated. HyFn display a wide spectrum of biological action both in vivo and in vitro.) [7] added to activated bicarbonate solutions to final concentrations of 10─9, 10─11 и 10─12 М. It can be seen that after initial elevation of PE intensity in all the samples it occasionally stabilized around certain quasi-stationary levels. HyFn in concentration of 10-12 M doubled PE intensity in comparison to the control samples, while in higher concentrations its effect of was much weaker. In other experiments HyFn exerted maximal stimulating effects in concentrations 10-15 and 10-19 M, while intermediate dilutions were less effective.
Such peculiar dose-dependence is characteristic of the effects of ultra-low doses of biologically active substances in different test-systems [8, 9].
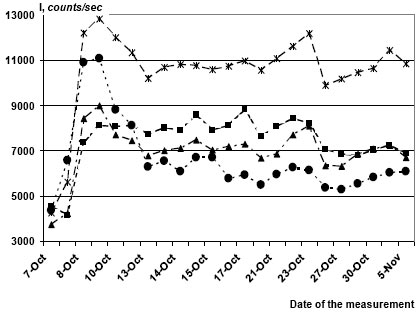
Fig. 1. Luminol-amplified PE from a series of sealed bicarbonate solutions (5 mM) with and without hydrated fullerenes C60 to which Н2О2 (0.01% final concentration) was added on October the 7th, 2008. Each curve represents a mean of intensity from triplicate samples: circles – control (no fullerenes); squares – 10-10 M, triangles – 10-11 M, asterisks – 10-12 M fullerenes. SD of the means for different series varied in the range between 3% (10-12 M fullerenes) and 8% (control).
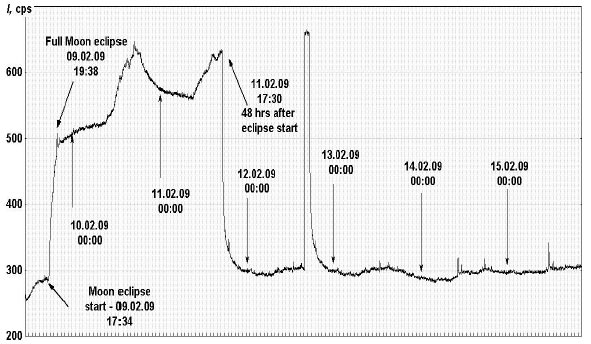
Fig. 2. Monitoring of PE from a sample with 5 mM bicarbonate solution to which H2O2 and Luminol were added on the 11th of November, 2008. The continuous session of PE monitoring has been started on the 26th of January, 2009. The record presented here started at 11:40 AM on the 9th of February. Original data was collected with 1 sec sampling time; the data presented here is moving average aggregation of consecutive 120 time points.
For more close studies of regularities of temporal variability of PE intensity single samples with bicarbonate solutions were fixed in a thermostatic sample compartment of the photon counter and continuous recording of PE from a sample was performed for several weeks without any interference into the process. Definite circadian rhythms of variations of PE with the minimum of PE around 7-9 hrs AM and maximum around 18-20 hrs PM was generally observed though waves of PE oscillations had different patterns on different days.
More or less regular circadian rhythms were drastically disturbed during the week started on February 9th. At this day the full Moon eclipse took place in Moscow. It started on February 9th at 17:34 PM and exactly at this moment of time PE intensity started to increase dramatically (Fig. 2). The Moon’s eclipse was full at 19:38 and a spike on the kinetic curve was observed just at this moment. After the Moon’s eclipse ended PE intensity did not decrease to its initial values, but started to oscillate with a circadian pattern at the level exceeding the previous one 2-3-fold. Surprisingly 48 hours after the start of the Moon’s eclipse photon emission rapidly declined to the level preceding the Moon’s eclipse. 3 days after the Moon’s eclipse PE intensity again raised more than 2-fold and 2 hours later it fall down to the initial level.
Currently it is difficult to suggest any hypothesis on the mechanism of action of the Moon’s eclipse on the activated bicarbonate solutions, though exact correlations between PE and time schedule of the Moon’s eclipse can hardly be explained by a mere coincidence.
One more indication of a highly excited state of activated bicarbonate solutions is that intensity of PE from the increased dramatically in response to addition of small portions (1:10 – 1:50) of non-activated bicarbonate solutions or even distilled water if the latter was previously agitated.
Thus, bicarbonate aqueous solutions activated with small quantities of H2O2 demonstrate the whole set of unusual properties related to their sustained ability to generate energy of electronic excitation despite the fact that they are restricted from matter exchange with their environment. Besides the system is able to accumulate high density energy as it can react to subtle irritations by strong and prolonged rising up of PE intensity. It is premature to suggest any more or less detailed model of processes that are responsible for permanently excited state of activated bicarbonate solution. However, some preconditions for such a model should be mentioned.
Aqueous systems as it is discussed above may be regarded as bi-phase: one phase being an electron donor, and the other is less organized and contains the oxidizer – oxygen. Carbonates present here may on the one hand support water structuring [10], increasing the potential difference between the two phases, and on the other – catalyze the process of water “burning” due to their ability to participate in free radical reactions. Thus, HCO3─ easily reduces one of the products of water splitting, hydroxyl radical (HO●), turning into carbonate СО3─●. The latter may support water oxidation, oxidize hydrogen peroxide [11], give rise to the emergence of organic compounds, such as oxocarbons [12]; the latter are able to originate cyclic red/ox reactions. A network of coupled and mutually supporting red/ox reactions emerges; yielding energy of electronic excitation. Thus (bi)carbonates may be regarded as peculiar catalysts of (reversible) water “burning”.
Regarding the role of H2O2 it should be stressed that combustion generally proceeds as the branching chain process [13]. Thus, it may start only when oxygen concentration exceeds a certain threshold, and only if it is initiated with a triggering stimulus (a “spark”) of high enough potential. When H2O2 is introduced into a bicarbonate solution it partially decomposes providing for a “spark” – a triggering impulse for the reaction initiation and supplying the system with additional oxygen to overcome the threshold needed for the kindling of chain reactions. After burning initiation energy released in its course promotes both fuel and oxygen excitation, resulting in the reinforcement of the burning process.
Whatever is the mechanism providing for the stable non-equilibrium state of bicarbonate aqueous systems its capability for permanent photon emission demands for the sustained energy supply. The only source of it under the given conditions is a thermal bath in which the system resides. As the structural temperature of organized water is lower than that of less organized water with which it is in contact, a temperature gradient between these two water phases exists. So organized water can constantly draw heat energy from the environment and transform it into energy of much higher density (potential) – energy of electronic excitation. From this it follows that bicarbonate solutions represent step-up energy transformers.
A significant part of water in living matter has the properties of a liquid crystalline phase. On the other hand, carbonates are the necessary constituent of living systems’ water. According to the concept presented here the suggested oxidation-reduction cycle of water (“water burning” = “water respiration”) catalyzed by carbonates is likely to be the fundamental source of energy for living systems, the basis of their residence in a highly excited state, their sensitivity to resonant external “irritants” of even very low intensity. On the whole intrinsic sustained activity of water containing carbonates is likely to be the necessary and sufficient condition for living systems emergence and existence.
REFERENCES
1. Szent-Gyorgyi A. Introduction to a supramolecular biology. NY & London: Academic Press, 1960, pp. 128-129.
2. Voeikov V.L. (2005). Cell Mol Biol., 51: 663-675.
3. Zheng J.M., Chin W.C., Khijniak E., et al. ( 2006). Adv Colloid Interface Sci., 23: 19-27
4. Henderson, Y. Adventures in Respiration. Modes of Asphyxiation and Methods of Resuscitation. Williams & Wilkins: Baltimore, 1938.
5. Medinas D.B., Cerchiaro G., Trindade D.F., et al. (2007). IUBMP Life, 59: 255-262
6. Voeikov V., et al. (2003) Clin. Lab., 49: 569.
7. Andrievsky G.V., et al. (2002). Chem. Phys. Letters., 364: 8-17.
8. Davenas E., et al. (1988). Nature, 333: 816-818.
9. Maltseva E.L., Palmina N.P., Burlakova E.B. (1998). Membrane & Cell Biology., 12: 251-268.
10. Pauling L. (1961) A molecular theory of general anesthesia. Science. 134, pp. 15-21
11. Komissarov G.G. Photosynthesis: Physical-chemical approach. Мoscow: URSS, 2003, p. 154-170.
12. Mazellier P., et al. (2002). New J. Chem., 26: 1784–1790.
13. Voeikov V.L., Naletov V.I. (1998). In: «Biophotons». Jiin-Ju Chang, Joachim Fisch, Fritz-Albert Popp. Kluwer Academic Publishers. Dortrecht, The Netherlands, рр. 93-108.
- Войдите или зарегистрируйтесь, чтобы получить возможность отправлять комментарии





 Нанотехнологии в водоочистке: достижения отечественной науки на службе здоровья.
Нанотехнологии в водоочистке: достижения отечественной науки на службе здоровья.
 Фильтры для воды "Геракл" с графеновым сорбентом: полный каталог и прайс-лист.
Фильтры для воды "Геракл" с графеновым сорбентом: полный каталог и прайс-лист.